Cookie preferences
This website uses cookies, which are necessary for the technical operation of the website and are always set. Other cookies, which increase the comfort when using this website, are used for direct advertising or to facilitate interaction with other websites and social networks, are only set with your consent.
Configuration
Technically required
These cookies are necessary for the basic functions of the shop.
"Allow all cookies" cookie
"Decline all cookies" cookie
CSRF token
Cookie preferences
Currency change
Customer-specific caching
FACT-Finder tracking
Individual prices
Selected shop
Session
Comfort functions
These cookies are used to make the shopping experience even more appealing, for example for the recognition of the visitor.
Note
Show the facebook fanpage in the right blod sidebar
Statistics & Tracking
Affiliate program
Conversion and usertracking via Google Tag Manager
Track device being used
| Item number | Size | Datasheet | Manual | SDS | Delivery time | Quantity | Price |
|---|---|---|---|---|---|---|---|
| A303-456A-T | 10 µl (2 µg) | - |
2 - 8 business days* |
164.00€
|
|||
| A303-456A | 100 µl | - |
2 - 8 business days* |
588.00€
|
If you have any questions, please use our Contact Form.
You can also order by e-mail: info@biomol.com
Larger quantity required? Request bulk
You can also order by e-mail: info@biomol.com
Larger quantity required? Request bulk
Protein function: E3 protein ligase that mediates ufmylation, the covalent attachment of the... more
Product information "Anti-UFL1"
Protein function: E3 protein ligase that mediates ufmylation, the covalent attachment of the ubiquitin-like modifier UFM1 to substrate proteins, a post-translational modification on lysine residues of proteins that may play a crucial role in a number of cellular processes. Mediates DDRGK1 ufmylation and may regulate the proteasomal degradation of DDRGK1 and CDK5RAP3 thereby modulating NF-kappa-B signaling (PubMed:20018847, PubMed:20164180, PubMed:20228063, PubMed:25219498). May also through TRIP4 ufmylation play a role in nuclear receptors-mediated transcription (PubMed:25219498). May play a role in the unfolded protein response, mediating the ufmylation of multiple proteins in response to endoplasmic reticulum stress (PubMed:23152784). [The UniProt Consortium]
| Keywords: | Anti-E3 UFM1-protein ligase 1, Anti-Novel LZAP-binding protein, Anti-E3 UFM1-protein transferase 1, Anti-Regulator of C53/LZAP and DDRGK1 |
| Supplier: | Bethyl Laboratories |
| Supplier-Nr: | A303-456A |
Properties
| Application: | WB, IP |
| Antibody Type: | Polyclonal |
| Conjugate: | No |
| Host: | Rabbit |
| Species reactivity: | human, mouse (Expected: bovine) |
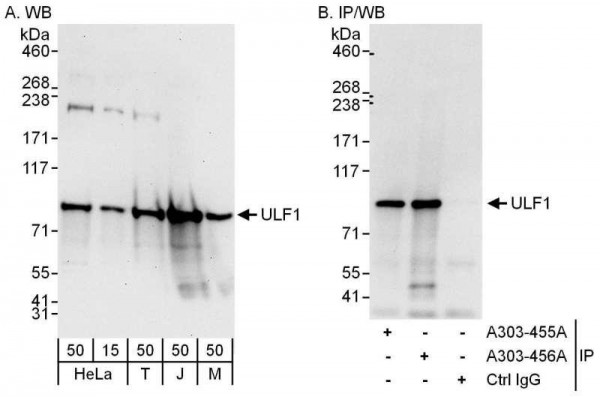
| Immunogen: | synthetic peptide. The epitope recognized by A303-456A-T maps to a region between residue 744 and 794 of human E3 UFM1-Protein Ligase 1 using the numbering given in entry NP_056138.1 (GeneID 23376). |
| Format: | Antigen Affinity Purified |
Database Information
| KEGG ID : | K22755 | Matching products |
| UniProt ID : | O94874 | Matching products |
| Gene ID | GeneID 23376 | Matching products |
Handling & Safety
| Storage: | +4°C |
| Shipping: | +4°C (International: +4°C) |
Caution
Our products are for laboratory research use only: Not for administration to humans!
Our products are for laboratory research use only: Not for administration to humans!
Information about the product reference will follow.
more
You will get a certificate here
Viewed