If you have any questions, please use our Contact Form.
You can also order by e-mail: info@biomol.com
Larger quantity required? Request bulk
You can also order by e-mail: info@biomol.com
Larger quantity required? Request bulk
Viewed
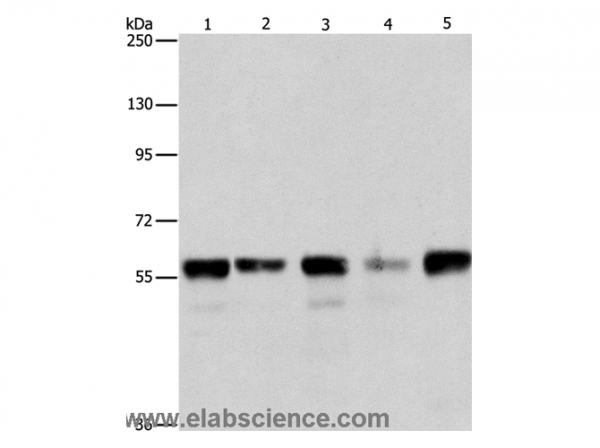
| Keywords: | Anti-ARI2, Anti-HT005, Anti-ARI-2, Anti-ARIH2, Anti-Triad1 protein, Anti-Protein ariadne-2 homolog, Anti-E3 ubiquitin-protein ligase ARIH2, Anti-RING-type E3 ubiquitin transferase ARIH2, ARIH2 Polyclonal Antibody |
| Supplier: | Elabscience |
| Supplier-Nr: | E-AB-12985 |
Properties
| Application: | WB, ELISA |
| Antibody Type: | Polyclonal |
| Conjugate: | No |
| Host: | Rabbit |
| Species reactivity: | human, mouse |
| Immunogen: | Synthetic peptide of human ARIH2 |
| Format: | Purified |
Database Information
| KEGG ID : | K11969 | Matching products |
| UniProt ID : | O95376 | Matching products |
| Gene ID | GeneID 10425 | Matching products |
Handling & Safety
| Storage: | -20°C |
| Shipping: | 4°C (International: -20°C) |
