Cookie preferences
This website uses cookies, which are necessary for the technical operation of the website and are always set. Other cookies, which increase the comfort when using this website, are used for direct advertising or to facilitate interaction with other websites and social networks, are only set with your consent.
Configuration
Technically required
These cookies are necessary for the basic functions of the shop.
"Allow all cookies" cookie
"Decline all cookies" cookie
CSRF token
Cookie preferences
Currency change
Customer-specific caching
FACT-Finder tracking
Individual prices
Selected shop
Session
Comfort functions
These cookies are used to make the shopping experience even more appealing, for example for the recognition of the visitor.
Note
Show the facebook fanpage in the right blod sidebar
Statistics & Tracking
Affiliate program
Conversion and usertracking via Google Tag Manager
Track device being used
If you have any questions, please use our Contact Form.
You can also order by e-mail: info@biomol.com
Larger quantity required? Request bulk
You can also order by e-mail: info@biomol.com
Larger quantity required? Request bulk
This gene is a member of the solute carrier family and encodes a cell surface, transmembrane... more
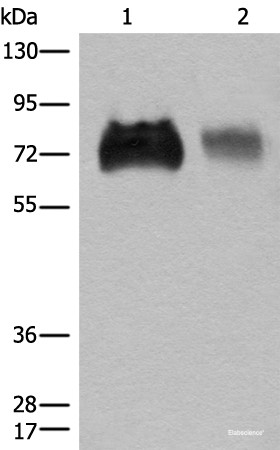
Product information "Anti-SLC3A2"
This gene is a member of the solute carrier family and encodes a cell surface, transmembrane protein. The protein exists as the heavy chain of a heterodimer, covalently bound through di-sulfide bonds to one of several possible light chains. The encoded transporter plays a role in regulation of intracellular calcium levels and transports L-type amino acids. Alternatively spliced transcript variants, encoding different isoforms, have been characterized. Protein function: Component of several heterodimeric amino acid transporter complexes (PubMed:11557028, PubMed:9829974, PubMed:9751058, PubMed:10391915, PubMed:10574970, PubMed:11311135). The precise substrate specificity depends on the other subunit in the heterodimer (PubMed:9829974, PubMed:9751058, PubMed:10391915, PubMed:10574970, PubMed:30867591, PubMed:10903140). The heterodimer with SLC3A2 functions as sodium-independent, high-affinity transporter that mediates uptake of large neutral amino acids such as phenylalanine, tyrosine, L-DOPA, leucine, histidine, methionine and tryptophan (PubMed:9751058, PubMed:11557028, PubMed:11311135, PubMed:11564694, PubMed:12117417, PubMed:12225859, PubMed:25998567, PubMed:30867591). The complexes with SLC7A6 and SLC7A7 mediate uptake of dibasic amino acids (PubMed:9829974, PubMed:10903140). The complexes function as amino acid exchangers (PubMed:11557028, PubMed:10903140, PubMed:12117417, PubMed:12225859, PubMed:30867591). Required for targeting of SLC7A5 and SLC7A8 to the plasma membrane and for channel activity (PubMed:9751058, PubMed:11311135, PubMed:30867591). Plays a role in nitric oxide synthesis in human umbilical vein endothelial cells (HUVECs) via transport of L-arginine. The heterodimer with SLC7A5/LAT1 may play a role in the transport of L-DOPA across the blood-brain barrier. May mediate blood-to-retina L- leucine transport across the inner blood-retinal barrier. The heterodimer with SLC7A5/LAT1 can mediate the transport of thyroid hormones triiodothyronine (T3) and thyroxine (T4) across the cell membrane (PubMed:11564694, PubMed:12225859). When associated with SLC7A5 or SLC7A8, involved in the cellular activity of small molecular weight nitrosothiols, via the stereoselective transport of L- nitrosocysteine (L-CNSO) across the transmembrane (PubMed:15769744). The heterodimer with SLC7A5 is involved in the uptake of toxic methylmercury (MeHg) when administered as the L-cysteine or D,L- homocysteine complexes (PubMed:12117417). Together with ICAM1, regulates the transport activity SLC7A8 in polarized intestinal cells, by generating and delivering intracellular signals. When associated with LAPTM4B, the heterodimer formed by SLC3A2 and SLC7A5 is recruited to lysosomes to promote leucine uptake into these organelles, and thereby mediates mTORC1 activation (PubMed:25998567). [The UniProt Consortium]
| Keywords: | Anti-CD98, Anti-MDU1, Anti-4F2hc, Anti-SLC3A2, Anti-4F2 heavy chain antigen, Anti-Solute carrier family 3 member 2, Anti-4F2 cell-surface antigen heavy chain, Anti-Lymphocyte activation antigen 4F2 large subunit, SLC3A2 Polyclonal Antibody |
| Supplier: | Elabscience |
| Supplier-Nr: | E-AB-18594 |
Properties
| Application: | WB, ELISA |
| Antibody Type: | Polyclonal |
| Conjugate: | No |
| Host: | Rabbit |
| Species reactivity: | human |
| Immunogen: | Fusion protein of human SLC3A2 |
Database Information
| KEGG ID : | K06519 | Matching products |
| UniProt ID : | P08195 | Matching products |
| Gene ID : | GeneID 6520 | Matching products |
Handling & Safety
| Storage: | -20°C |
| Shipping: | 4°C (International: -20°C) |
Caution
Our products are for laboratory research use only: Not for administration to humans!
Our products are for laboratory research use only: Not for administration to humans!
Information about the product reference will follow.
more
You will get a certificate here
Viewed




