Cookie preferences
This website uses cookies, which are necessary for the technical operation of the website and are always set. Other cookies, which increase the comfort when using this website, are used for direct advertising or to facilitate interaction with other websites and social networks, are only set with your consent.
Configuration
Technically required
These cookies are necessary for the basic functions of the shop.
"Allow all cookies" cookie
"Decline all cookies" cookie
CSRF token
Cookie preferences
Currency change
Customer-specific caching
FACT-Finder tracking
Individual prices
Selected shop
Session
Comfort functions
These cookies are used to make the shopping experience even more appealing, for example for the recognition of the visitor.
Note
Show the facebook fanpage in the right blod sidebar
Statistics & Tracking
Affiliate program
Conversion and usertracking via Google Tag Manager
Track device being used
| Item number | Size | Datasheet | Manual | SDS | Delivery time | Quantity | Price |
|---|---|---|---|---|---|---|---|
| NSJ-FY13350 | 100 µg | - | - |
3 - 10 business days* |
790.00€
|
If you have any questions, please use our Contact Form.
You can also order by e-mail: info@biomol.com
Larger quantity required? Request bulk
You can also order by e-mail: info@biomol.com
Larger quantity required? Request bulk
Adding 0.2 ml of distilled water will yield a concentration of 500 ug/ml. GCNT2 antibody detects... more
Product information "Anti-GCNT2 / Beta-1,6-N-acetylglucosaminyltransferase 2"
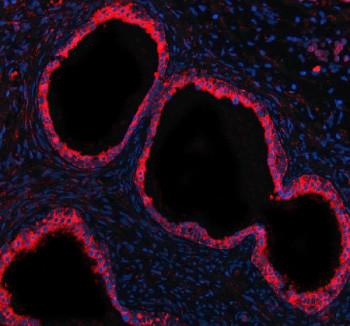
Adding 0.2 ml of distilled water will yield a concentration of 500 ug/ml. GCNT2 antibody detects Beta-1,6-N-acetylglucosaminyltransferase 2, a Golgi-localized glycosyltransferase encoded by the GCNT2 gene on chromosome 6p24.3. GCNT2 is an enzyme that catalyzes the branching of poly-N-acetyllactosamine chains on glycoproteins and glycolipids, forming the I and i blood group antigens. It belongs to the glycosyltransferase 14 family and plays an essential role in the biosynthesis of complex glycans, particularly those expressed on erythrocytes, epithelial cells, and the ocular lens. GCNT2 expression is highest in red blood cells, corneal tissue, and gastrointestinal epithelium, where it contributes to cell adhesion and recognition processes.GCNT2 functions by transferring N-acetylglucosamine residues via a beta1,6 linkage to galactose units, generating branched carbohydrate structures. These modifications affect protein stability, receptor signaling, and cell-cell interactions. The enzyme has three transcript isoforms (A, B, and C), each exhibiting tissue-specific expression patterns and promoter usage. GCNT2A is predominant in erythroid cells, while GCNT2B and GCNT2C are expressed in non-hematopoietic tissues such as the lens and gastrointestinal tract.Structurally, GCNT2 contains a conserved catalytic domain typical of glycosyltransferases, including the DXH motif required for donor sugar binding. It is anchored in the Golgi membrane by a short N-terminal transmembrane domain, positioning its catalytic site within the lumen for glycan modification. GCNT2 belongs to the GT14 family of glycosyltransferases, which also includes GCNT1 and GCNT3, enzymes responsible for branching of mucin-type O-glycans and other carbohydrate chains.Functionally, GCNT2 is responsible for converting linear i antigens into branched I antigens during erythrocyte maturation. Loss of GCNT2 activity results in the adult i blood group phenotype, characterized by persistence of fetal-type linear glycans. In the lens, GCNT2 participates in glycoprotein processing required for lens transparency, while in epithelial tissues, it regulates mucin glycosylation that influences cell adhesion and immune defense. Known substrates of GCNT2 include glycoproteins bearing poly-N-acetyllactosamine extensions such as laminin and selectins.Mutations in GCNT2 are associated with congenital cataracts and the rare adult i blood group phenotype. Reduced enzymatic activity leads to accumulation of unbranched glycan structures, affecting membrane organization and cell surface recognition. In cancer, altered GCNT2 expression has been observed in breast, colon, and gastric carcinomas, where changes in glycan branching influence cell motility and metastasis. Pathway associations include glycosphingolipid biosynthesis, protein glycosylation, and immune recognition processes.Immunohistochemical staining using GCNT2 antibody shows Golgi localization in epithelial and erythroid cells. The GCNT2 antibody from NSJ Bioreagents is an effective tool for investigating glycan biosynthesis, erythrocyte differentiation, and cancer-related changes in glycosylation patterns.
| Keywords: | Anti-IGNT, Anti-GCNT2, Anti-I-branching enzyme, Anti-N-acetyllactosaminide beta-1,6-N-acetylglucosaminyl-transferase, GCNT2 Antibody / Beta-1,6-N-acetylglucosaminyltransferase 2 |
| Supplier: | NSJ Bioreagents |
| Supplier-Nr: | FY13350 |
Properties
| Application: | WB, IHC, ICC/IF, IF, FC, ELISA |
| Antibody Type: | Polyclonal |
| Conjugate: | No |
| Host: | Rabbit |
| Species reactivity: | human, mouse, rat |
| Immunogen: | E.coli-derived human GCNT2 recombinant protein (Position: E27-Q397) |
| Format: | Purified |
Database Information
| KEGG ID : | K00742 | Matching products |
| UniProt ID : | Q8N0V5 | Matching products |
| Gene ID : | GeneID 2651 | Matching products |
Handling & Safety
| Storage: | +4°C |
| Shipping: | +4°C (International: +4°C) |
Caution
Our products are for laboratory research use only: Not for administration to humans!
Our products are for laboratory research use only: Not for administration to humans!
Information about the product reference will follow.
more
You will get a certificate here
Viewed