Cookie preferences
This website uses cookies, which are necessary for the technical operation of the website and are always set. Other cookies, which increase the comfort when using this website, are used for direct advertising or to facilitate interaction with other websites and social networks, are only set with your consent.
Configuration
Technically required
These cookies are necessary for the basic functions of the shop.
"Allow all cookies" cookie
"Decline all cookies" cookie
CSRF token
Cookie preferences
Currency change
Customer-specific caching
FACT-Finder tracking
Individual prices
Selected shop
Session
Comfort functions
These cookies are used to make the shopping experience even more appealing, for example for the recognition of the visitor.
Note
Show the facebook fanpage in the right blod sidebar
Statistics & Tracking
Affiliate program
Conversion and usertracking via Google Tag Manager
Track device being used
If you have any questions, please use our Contact Form.
You can also order by e-mail: info@biomol.com
Larger quantity required? Request bulk
You can also order by e-mail: info@biomol.com
Larger quantity required? Request bulk
The naturally-occurring estrogens are estrone (E1, Cay-10006485), estradiol (E2, Cay-10006315),... more
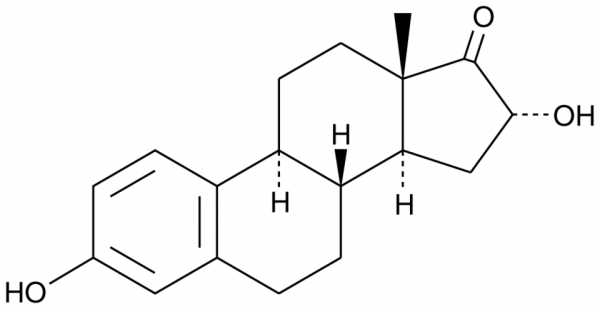
Product information "16alpha-hydroxy Estrone"
The naturally-occurring estrogens are estrone (E1, Cay-10006485), estradiol (E2, Cay-10006315), and estriol (E3, Cay-10006484). 16alpha-hydroxy Estrone (16alpha-OHE1) is a hydroxylated metabolite of E1 as well as an interconversion product with E2. E1 is 16alpha-hydroxylated by cytochrome P450 (CYP) isoforms, including CYP1A1, CYP3A5, CYP3A4, and CYP3A7, with CYP3A5 being breast-specific. 16alpha-OHE1 is sulphatized or glucuronidated before excretion. It is increased in rheumatoid arthritis and decreased by physical activity. Unlike the parent estrogens and other hydroxylated metabolites of E1, 16alpha-OHE1 binds covalently and persistently activates estrogen receptors. In addition, this metabolite increases cell proliferation and does not suppress TNF-alpha secretion, whereas other estrogen metabolites are not pro-proliferative and have marked effects on TNF-alpha secretion. The levels of 16alpha-OHE1 are increased in some forms of hormone therapy. Because hormone therapy increases breast cancer risk, 16alpha-OHE1 has been implicated as a risk factor for breast cancer, although supportive data remains elusive.Formal Name: 3,16alpha-dihydroxy-estra-1,3,5(10)-trien-17-one. CAS Number: 566-76-7. Synonyms: 16alpha-hydroxy E1, 16alpha-OHE1. Molecular Formula: C18H22O3. Formula Weight: 286.4. Purity: >98%. Formulation: (Request formulation change), A crystalline solid. Solubility: DMF: 30 mg/ml, DMF:PBS (pH 7.2) (1:5): 0.15 mg/ml, DMSO: 20 mg/ml. lambdamax: 281 nm. SMILES: O=C1[C@H](O)C[C@@]2([H])[C@]3([H])CCC4=CC(O)=CC=C4[C@@]3([H])CC[C@@]21C. InChi Code: InChI=1S/C18H22O3/c1-18-7-6-13-12-5-3-11(19)8-10(12)2-4-14(13)15(18)9-16(20)17(18)21/h3,5,8,13-16,19-20H,2,4,6-7,9H2,1H3/t13-,14-,15+,16-,18+/m1/s1. InChi Key: WPOCIZJTELRQMF-QFXBJFAPSA-N.
| Keywords: | 16alpha-OHE1, 3,16alpha-dihydroxy-estra-1,3,5(10)-trien-17-one |
| Supplier: | Cayman Chemical |
| Supplier-Nr: | 15208 |
Properties
| Application: | Estrogen receptor agonist, Increases cell proliferation |
| MW: | 286.4 D |
| Formula: | C18H22O3 |
| Purity: | >98% |
| Format: | Crystalline Solid |
Database Information
| CAS : | 566-76-7| Matching products |
Handling & Safety
| Storage: | -20°C |
| Shipping: | +20°C (International: -20°C) |
| Signal Word: | Warning |
| GHS Hazard Pictograms: |
|
| H Phrases: | H351 |
| P Phrases: | P201, P202, P280, P308+313, P405, P501 |
Caution
Our products are for laboratory research use only: Not for administration to humans!
Our products are for laboratory research use only: Not for administration to humans!
Information about the product reference will follow.
more
You will get a certificate here
Viewed


![16alpha-Hydroxyestrone-[2,3,4-13C3] 16alpha-Hydroxyestrone-[2,3,4-13C3]](/custom/plugins/NetiThemeBiomol/Resources/Themes/Frontend/Biomol/frontend/_public/src/img/no-picture.jpg)