Cookie preferences
This website uses cookies, which are necessary for the technical operation of the website and are always set. Other cookies, which increase the comfort when using this website, are used for direct advertising or to facilitate interaction with other websites and social networks, are only set with your consent.
Configuration
Technically required
These cookies are necessary for the basic functions of the shop.
"Allow all cookies" cookie
"Decline all cookies" cookie
CSRF token
Cookie preferences
Currency change
Customer-specific caching
FACT-Finder tracking
Individual prices
Selected shop
Session
Comfort functions
These cookies are used to make the shopping experience even more appealing, for example for the recognition of the visitor.
Note
Show the facebook fanpage in the right blod sidebar
Statistics & Tracking
Affiliate program
Conversion and usertracking via Google Tag Manager
Track device being used
If you have any questions, please use our Contact Form.
You can also order by e-mail: info@biomol.com
Larger quantity required? Request bulk
You can also order by e-mail: info@biomol.com
Larger quantity required? Request bulk
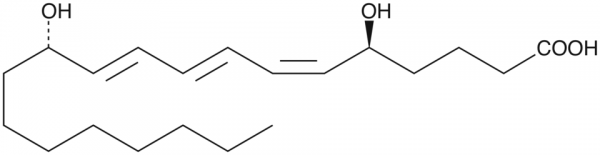
12-epi LTB3 is an LTB isomer that would not be formed either by known enzymatic or non-enzymatic... more
Product information "12-epi Leukotriene B3"
12-epi LTB3 is an LTB isomer that would not be formed either by known enzymatic or non-enzymatic processes. The conversion of LTA into different isomers of LTB is complex, and has generated confusion in the past. The enzymatic hydrolysis of LTA leads to LTB, which is the corresponding 5(S),12(R)-dihydroxy acid containing a 6(Z),8(E),10(E) conjugated triene. Non-enzymatic hydrolysis of LTA produces none of this material, but instead produces both the 12(S) and the 12(R) isomers of LTB in which the triene component now contains a 6-trans double bond. 12-epi (or 12(S)) LTB isomers which contain the natural 6-cis olefin are not produced either by enzymatic or by non-enzymatic processes. Further, 3-series LTs (derived from Mead acid) are only seen in essential fatty acid deficiency. Mead acid is a good substrate for 5-lipoxygenase, leading to LTA3. Enzymatic conversion of LTA3 to LTB3 is very low because LTA3 is actually an inhibitor of LTA hydrolase. Non-enzymatic hydrolysis of LTA3 leads to 6-trans-12-epi LTB3, and to 6-trans LTB3. The biological activity of 12-epi LTB3 has not been explored.Formal Name: 5S,12S-dihydroxy-6Z,8E,10E-eicosatrienoic acid. Synonyms: 12-epi LTB3. Molecular Formula: C20H34O4. Formula Weight: 338.5. Purity: >97%. Formulation: (Request formulation change), A solution in ethanol. Solubility: DMF: Miscible, DMSO: Miscible, Ethanol: Miscible, PBS (pH 7.2): 1 mg/ml (from 13-HODE). lambdamax: 270 nm. SMILES: CCCCCCCC[C@H](O)/C=C/C=C/C=C\[C@@H](O)CCCC(O)=O. InChi Code: InChI=1S/C20H34O4/c1-2-3-4-5-6-9-13-18(21)14-10-7-8-11-15-19(22)16-12-17-20(23)24/h7-8,10-11,14-15,18-19,21-22H,2-6,9,12-13,16-17H2,1H3,(H,23,24)/b8-7+,14-10+,15-11-/t18-,19+/m0/s1. InChi Key: NGTXCORNXNELNU-OUZMKVEGSA-N.
| Keywords: | 12-epi LTB3, 5S,12S-dihydroxy-6Z,8E,10E-eicosatrienoic acid |
| Supplier: | Cayman Chemical |
| Supplier-Nr: | 20134 |
Properties
| Application: | Bioactive lipid assays |
| MW: | 338.5 D |
| Formula: | C20H34O4 |
| Purity: | >97% |
| Format: | Solution |
Database Information
Handling & Safety
| Storage: | -20°C |
| Shipping: | -20°C (International: -20°C) |
| Signal Word: | Danger |
| GHS Hazard Pictograms: |
|
| H Phrases: | H225 |
| P Phrases: | P210, P233, P240, P241, P242, P243, P280, P303+361+353, P370+378, P403+235, P501 |
Caution
Our products are for laboratory research use only: Not for administration to humans!
Our products are for laboratory research use only: Not for administration to humans!
Information about the product reference will follow.
more
You will get a certificate here
Viewed