Obesity represents one of the biggest global health problems. To date, lifestyle changes (diet and exercise) have been a major part of the treatment of obesity. With the addition of the diabetes drug semaglutide, a pharmacological therapy option could now be used to effectively reduce weight, a new study suggests.
Hope for the Treatment of Obesity
Semaglutide is an active ingredient approved as a treatment for type 2 diabetes in the EU in 2018. It is a glucagon-like peptide (GLP-1) agonist, which binds to the GLP-1 receptor. Binding of semaglutide to this receptor results in several pharmacological effects that are important in the treatment of diabetes. These include promoting glucose-dependent insulin secretion by pancreatic beta cells, decreasing gluconeogenesis by the liver, and slowing gastric emptying [1]. During clinical trials of semaglutide in patients with type 2 diabetes, it was noticed that patients showed a significant reduction in body weight after injection with the drug in combination with changes in relevant lifestyle habits (diet and exercise).
New Study Tests Weight Loss With Semaglutide in Non-Diabetics
To examine weight loss with semaglutide in nondiabetics, Kushner et al. [2] conducted a double-blind study of 1961 adult participants. Subjects had either a body mass index of 30 and above or at least 27 with weight-related comorbidities, not including diabetes. Study participants were divided in a 2:1 ratio and received 2.5 mg of semaglutide subcutaneously, or the placebo, once weekly for 68 weeks. Additionaly, lifestyle changes were made (500kcal less per day and 150 minutes of physical activity such as walking). After 68 weeks, there was a break without the administration of semaglutide and without the lifestyle changes.
Semaglutide Was Shown to Help Reduce Body Weight Even in Non-Diabetics
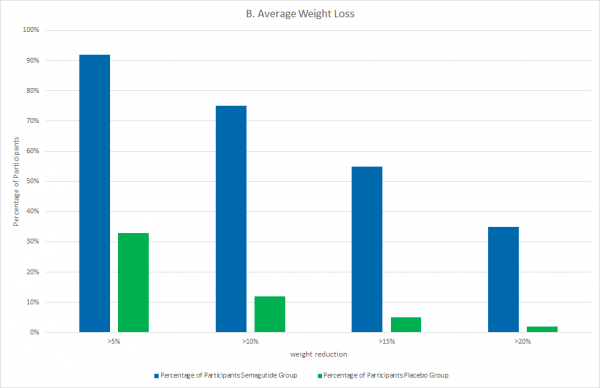
In the semaglutide group, participants maintained a continuous weight loss from week 4 onwards. The maximum weight reduction was measured after 60 weeks. The average weight loss of participants after the study duration of 68 weeks was 14.9% in the semaglutide group and 2.4% in the placebo group. A weight reduction of at least 5% was achieved by 92.4% of participants in the semaglutide group, whereas only 33.1% in the placebo group achieved the same reduction. Other health parameters also improved with the administration of the drug. For example, hip circumference decreased by an average of 13.54 cm (placebo group average 4.13 cm), blood pressure decreased by 6.16 mmHg (placebo 1.60 mmHg), and blood lipid levels improved. The SF-36 physical function score also increased significantly in the semaglutide group. This is a patient survey of health-related well-being that includes factors such as pain, general body functions, and mental health. The weight loss achieved with semaglutide in combination with changes in lifestyle habits was found to improve many health-related parameters and increase the general well-being of the participants.

Fig.1: The average weight loss of the participants after the study duration of 68 weeks was 14.9% in the semaglutide group and 2.4% in the placebo group. Adapted from Kushner et al.

Fig.2: A weight reduction of at least 5% was achieved by 92.4% of participants from the semaglutide group whereas only 33.1% from the placebo group achieved the same reduction. The proportion of greater weight loss was also significantly higher in the semaglutide group (e.g., weight reduction of more than 20% in nearly 35% subjects compared with 2% in the control group). Adapted from Kushner et al.
Only Few Side Effects So Far
Compared with the improvement in multiple parameters (blood lipid levels, hypertension, etc.), few negative side effects have been observed in the study by Kushner et al. The most common were gastrointestinal symptoms such as nausea and vomiting. These symptoms were predominantly mild. More severe side effects occurred in 9.8% of subjects (compared to 6.4% in the control group). These side effects are already known from previous studies. A special feature of the drug is its half-life. Semaglutide only needs to be injected subcutaneously once a week to achieve the desired effect. However, to obtain the positive effect studied by Kushner et al. the drug would most likely need to be applied throughout the patient´s life.
Further Obesity Research Needed
A reliable and effective reduction of body weight is the main goal in the treatment of obesity. To date, there are few drugs that can effectively help with weight loss. The research approaches to adressing this need are diverse. While a large branch of research focuses on the genetic component of obesity, intensive research is being done in the field of adipogenesis. The different phases of the cell cycle, as well as specific growth factors and inhibitors represent potential targets for active compounds. The intersection between obesity/cancer and obesity/inflammatory disease research is also of great importance. It is estimated that approximately 1.9 billion adults worldwide suffer from obesity. In the EU alone, up to 61.5% of the population is likely to be overweight, according to Eurostat data [3]. High levels of obesity and the associated consequences pose both a personal and a public health risk underscoring the need for further research.
References
- Semaglutid - Anwendung, Wirkung, Nebenwirkungen | Gelbe Liste (gelbe-liste.de)
- Kushner et al., Once-Weekly Semaglutide in Adults with Overweight or Obesity, March 18, 2021, N Engl J Med 2021; 384:989-1002
- Statistik Adipositas EU- Adipositas: EU-Forschung sagt Übergewicht den Kampf an | Research*eu Magazine | Issue 84 | CORDIS | European Commission (europa.eu)