Cookie preferences
This website uses cookies, which are necessary for the technical operation of the website and are always set. Other cookies, which increase the comfort when using this website, are used for direct advertising or to facilitate interaction with other websites and social networks, are only set with your consent.
Configuration
Technically required
These cookies are necessary for the basic functions of the shop.
"Allow all cookies" cookie
"Decline all cookies" cookie
CSRF token
Cookie preferences
Currency change
Customer-specific caching
FACT-Finder tracking
Individual prices
Selected shop
Session
Comfort functions
These cookies are used to make the shopping experience even more appealing, for example for the recognition of the visitor.
Note
Show the facebook fanpage in the right blod sidebar
Statistics & Tracking
Affiliate program
Conversion and usertracking via Google Tag Manager
Track device being used
| Item number | Size | Datasheet | Manual | SDS | Delivery time | Quantity | Price |
|---|---|---|---|---|---|---|---|
| AG-45B-0011-KI01 | 96 wells |
3 - 9 business days* |
660.00€
|
If you have any questions, please use our Contact Form.
You can also order by e-mail: info@biomol.com
Larger quantity required? Request bulk
You can also order by e-mail: info@biomol.com
Larger quantity required? Request bulk
Detects full-length and cleaved mouse Gasdermin D (C-terminus) in cell culture supernatants and... more
Product information "Gasdermin D (mouse) ELISA Kit"
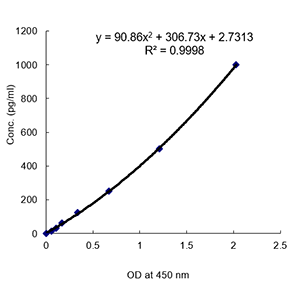
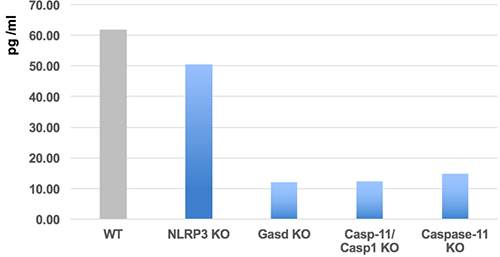
Detects full-length and cleaved mouse Gasdermin D (C-terminus) in cell culture supernatants and cell extracts. Does not cross-react with human gasdermin D. Sensitivity: 14pg/ml. Range: 15.6 to 1000pg/ml. Protein function: Gasdermin-D, N-terminal: Promotes pyroptosis in response to microbial infection and danger signals. Produced by the cleavage of gasdermin-D by inflammatory caspases CASP1 or CASP4 in response to canonical, as well as non-canonical (such as cytosolic LPS) inflammasome activators (PubMed:26611636, PubMed:26375259, PubMed:26375003, PubMed:27418190, PubMed:27385778, PubMed:27383986). After cleavage, moves to the plasma membrane where it strongly binds to membrane inner leaflet lipids, including monophosphorylated phosphatidylinositols, such as phosphatidylinositol 4-phosphate, bisphosphorylated phosphatidylinositols, such as phosphatidylinositol (4,5)- bisphosphate, as well as phosphatidylinositol (3,4,5)- trisphosphate, and more weakly to phosphatidic acid and phosphatidylserine. Homooligomerizes within the membrane and forms pores of 10 - 15 nanometers (nm) of inner diameter, allowing the release of mature IL1B and triggering pyroptosis. Exhibits bactericidal activity. Gasdermin-D, N-terminal released from pyroptotic cells into the extracellular milieu rapidly binds to and kills both Gram-negative and Gram-positive bacteria, without harming neighboring mammalian cells, as it does not disrupt the plasma membrane from the outside due to lipid-binding specificity. Under cell culture conditions, also active against intracellular bacteria, such as Listeria monocytogenes. Strongly binds to bacterial and mitochondrial lipids, including cardiolipin. Does not bind to phosphatidylethanolamine or phosphatidylcholine (PubMed:27383986). [The UniProt Consortium] Inflammasomes are multimeric protein complexes that comprise a sensor (e.g. NLRP3), an adaptor (ASC/Pycard) and the procaspase-1. An inflammasome assembles in response to a diverse range of pathogen-associated or danger-associated molecular patterns (PAMPs or DAMPs). The inflammasome platform leads to activation of caspase-1, which further induces maturation of interleukin-1beta and -18 (IL-1beta and IL-18) through proteolytic cleavage of pro-IL-1beta and pro-IL-18. Activated caspase-1, and also the recently characterized caspase-11 non-canonical inflammasome pathway, also cleave the intracellular gasdermin D, which leads to a particular form of inflammatory cell death called pyroptosis. The gasdermin family members contain N-terminal domains that are capable of forming membrane pores to induce cytolysis, whereas the C-terminal domains of gasdermins function as inhibitors of such cytolysis through intramolecular domain association. Caspase-1 or -11 cleavage of gasdermin D is required for regulation of pyroptosis: upon protease cleavage of the gasdermin N- and C-domain linker, the disruption of the intramolecular domain interaction in the presence of lipids releases the N-domain to assemble oligomeric membrane pores that trigger cell death. Gasdermin D seems to be a key effector in the LPS-induced lethal sepsis.
| Keywords: | Gsdmd, Gsdmdc1, Gasdermin Domain-containing Protein 1, Gasdermin-D (C-terminal), GSDMD-CT |
| Supplier: | AdipoGen Life Sciences |
| Supplier-Nr: | AG-45B-0011 |
Properties
| Application: | ELISA |
| Species reactivity: | mouse |
| Format: | Solid Phase |
Database Information
| KEGG ID : | K20917 | Matching products |
| UniProt ID : | Q9D8T2 | Matching products |
| Gene ID : | GeneID 69146 | Matching products |
Handling & Safety
| Storage: | +4°C |
| Shipping: | +4°C (International: +4°C) |
Caution
Our products are for laboratory research use only: Not for administration to humans!
Our products are for laboratory research use only: Not for administration to humans!
Information about the product reference will follow.
more
You will get a certificate here
Viewed