Cerebrosides
Cerebrosides (galactosylceramides) are primarily found in neuronal tissues and are the major glycosphingolipids in the central nervous system. They are the largest single component of the myelin sheath of nerves and seem to act, along with other molecules, to form part of the structural support of the myelin sheath.1 Cerebrosides are involved in a very wide range of biological activities such as cell agglutination, intracellular communication, cellular development, and have antitumor/cytotoxic effects.2 Galactocerebroside can be metabolized into sulfatide which is also abundant in the nervous system and myelin sheaths. Due to the relatively high melting point of cerebrosides (much greater than physiological body temperature), they have a paracrystalline structure. Krabbe disease (globoid cell leukodystrophy) is characterized by a deficiency in the enzyme galactocerebrosidase, which is responsible for degrading galactocerebroside. This leads to an accumulation of cerebroside and psychosine, which is very cytotoxic and can result in demyelination of nerves and loss of axonal conductivity.3
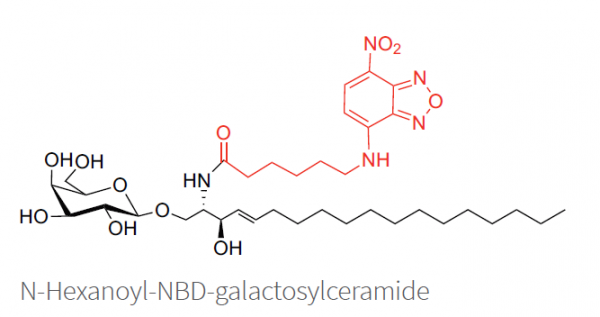
NBD-labeling
NBD-galactosylceramide is a fluorescent cerebroside with an excitation/emission maximum of ~466/539 nm. NBD (7-nitrobenz-2-oxa-1,3-diazol-4-yl) has long been one of the most common fluorescent lipid labels. Studies have shown that NBD-labeled lipids mimic their natural lipid counterparts very well in most applications.4 The NBD group demonstrates only a small influence on lipid adsorption into cells and cellular membranes, especially when the fatty acid is a short chain. This fluorescent analog is generally comparable to galactosylceramide in many biological functions.
Lissamine-rhodamine B-dodecanoyl-galactosylceramide
Lissamine-rhodamine B-dodecanoyl-galactosylceramide is a fluorescent-labeled glycosphingolipid containing a lissamine-rhodamine B marker attached to the sphingosine. This fluorescent standard from Matreya is excellent for use in the identification and isolation of cerebrosides in Krabbe disease and other studies. Lissamine-rhodamine B dyes have an excitation/emission maximum of ~560/580 nm. The fluorescent marker is attached via a 12-carbon linker reducing the interaction of the fluorophore with the sphingolipid.
References:
- S.L. Miller and L. Denisova. Cycloserine-induced decrease of cerebroside in myelin. Lipids 33(4), 441-443 (1998).
- X. Zhou, L. Tang, and Y. Liu (2009) An Isomeric Mixture of Novel Cerebrosides Isolated from Impatiens pritzellii Reduces Lipopolysaccharide-Induced Release of IL-18 from Human Peripheral Blood Mononuclear Cells. Lipids, Vol. 44:8 pp. 759-763
- X. Han and H. Cheng (2005) Characterization and direct quantitation of cerebroside molecular species from lipid extracts by shotgun lipidomics. Journal of Lipid Research, Vol.46:1 pp. 163-175
- A. Chattopadhyay (1990) Chemistry and biology of N-(7-nitrobenz-2-oxa-1,3-diazol-4-yl)-labeled lipids: fluorescent probes of biological and model membranes. Chemistry and Physics of Lipids, Vol. 53:1 pp. 1-15