Cookie preferences
This website uses cookies, which are necessary for the technical operation of the website and are always set. Other cookies, which increase the comfort when using this website, are used for direct advertising or to facilitate interaction with other websites and social networks, are only set with your consent.
Configuration
Technically required
These cookies are necessary for the basic functions of the shop.
"Allow all cookies" cookie
"Decline all cookies" cookie
CSRF token
Cookie preferences
Currency change
Customer-specific caching
FACT-Finder tracking
Individual prices
Selected shop
Session
Comfort functions
These cookies are used to make the shopping experience even more appealing, for example for the recognition of the visitor.
Note
Show the facebook fanpage in the right blod sidebar
Statistics & Tracking
Affiliate program
Conversion and usertracking via Google Tag Manager
Track device being used
| Item number | Size | Datasheet | Manual | SDS | Delivery time | Quantity | Price |
|---|---|---|---|---|---|---|---|
| Cay38017-1 | 1 U | - |
6 - 10 business days* |
223.00€
|
If you have any questions, please use our Contact Form.
You can also order by e-mail: info@biomol.com
Larger quantity required? Request bulk
You can also order by e-mail: info@biomol.com
Larger quantity required? Request bulk
Xanthine oxidase (XO) is a complex metalloflavoprotein involved in purine catabolism, oxidative... more
Product information "Xanthine Oxidase (bovine)"
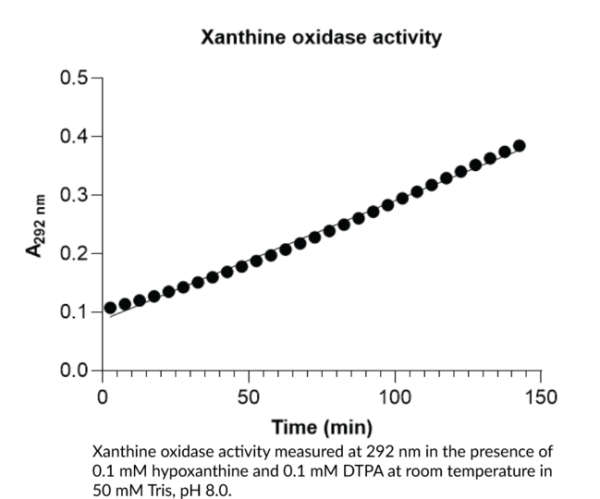
Xanthine oxidase (XO) is a complex metalloflavoprotein involved in purine catabolism, oxidative stress, and xenobiotic metabolism, among other processes. It exists as a homodimer and each monomer is composed of an N-terminal domain that contains two unequal iron-sulfur clusters, an intermediate domain that contains the FAD cofactor, and a C-terminal domain that contains the molybdopterin cofactor. XO is primarily expressed in the liver and intestines, has been found in blood, serum, and milk, and localizes to the cytosol, cell membrane, and peroxisomes.1 It is produced by irreversible or reversible post-translational modification of xanthine dehydrogenase (XD) via limited proteolysis or oxidation of XD thiol groups, respectively. XO catalyzes the conversion of hypoxanthine to xanthine to uric acid and uses molecular oxygen as the electron acceptor, compared with XD that uses NAD+, and this results in the generation of hydrogen peroxide and superoxide, which can react with nitric oxide (NO) to produce peroxynitrite. In hypoxic conditions, XO converts nitrates and nitrite to NO. Increased blood levels of XO's enzymatic product uric acid, hyperuricemia, is associated with several diseases, including gout, cardiovascular disease, ischemia-reperfusion injury, obesity, and diabetes. XO has commonly been used in coupled enzyme assays to measure superoxide dismutase (SOD) activity. Cayman's Xanthine Oxidase (bovine) protein can be used for enzyme activity assays. Protein function: Key enzyme in purine degradation. Catalyzes the oxidation of hypoxanthine to xanthine. Catalyzes the oxidation of xanthine to uric acid. Contributes to the generation of reactive oxygen species. [The UniProt Consortium]
| Keywords: | XD, XO, XDH, XOR, Xanthine oxidase, Xanthine dehydrogenase, Xanthine oxidoreductase, Xanthine dehydrogenase/oxidase |
| Supplier: | Cayman Chemical |
| Supplier-Nr: | 38017 |
Properties
| Application: | Active enzyme |
| Conjugate: | No |
| Host: | bovine milk |
| Species reactivity: | bovine |
| MW: | 146.79 kD |
| Format: | Ammonium Sulfate Precipitated |
Database Information
| KEGG ID : | K00106 | Matching products |
| UniProt ID : | P80457 | Matching products |
| Gene ID : | GeneID 280960 | Matching products |
Handling & Safety
| Storage: | +4°C |
| Shipping: | +20°C (International: +4°C) |
Caution
Our products are for laboratory research use only: Not for administration to humans!
Our products are for laboratory research use only: Not for administration to humans!
Information about the product reference will follow.
more
You will get a certificate here
Viewed

