Cookie preferences
This website uses cookies, which are necessary for the technical operation of the website and are always set. Other cookies, which increase the comfort when using this website, are used for direct advertising or to facilitate interaction with other websites and social networks, are only set with your consent.
Configuration
Technically required
These cookies are necessary for the basic functions of the shop.
"Allow all cookies" cookie
"Decline all cookies" cookie
CSRF token
Cookie preferences
Currency change
Customer-specific caching
FACT-Finder tracking
Individual prices
Selected shop
Session
Comfort functions
These cookies are used to make the shopping experience even more appealing, for example for the recognition of the visitor.
Note
Show the facebook fanpage in the right blod sidebar
Statistics & Tracking
Affiliate program
Conversion and usertracking via Google Tag Manager
Track device being used
If you have any questions, please use our Contact Form.
You can also order by e-mail: info@biomol.com
Larger quantity required? Request bulk
You can also order by e-mail: info@biomol.com
Larger quantity required? Request bulk
Pralidoxime reactivates acetylcholinesterase (AChE) that has been deactivated by binding of... more
Product information "Pralidoxime (chloride)"
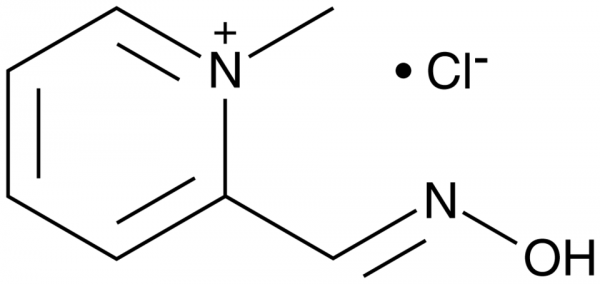
Pralidoxime reactivates acetylcholinesterase (AChE) that has been deactivated by binding of organophosphates to its esteratic site. Pralidoxime binds to the anionic site of AChE and displaces the phosphate from the esteratic site through formation of phosphate-pralidoxime conjugates. It reactivates paraoxon- and diisopropyl fluorophosphate-inactivated human red blood cell (RBC) AChE with IC50 shifts of 0.3 and 0.8 nM per µM of pralidoxime, respectively., At a concentration of 10 µM, it reactivates human RBC AChE that has been inactivated by chlorpyrifos (Cay-21412), diazinon (Cay-23769), and malathion (Cay-22998) by 17, 61, and 36%, respectively. Pralidoxime binds to sarin-bound hAChE (Kd = 25.72 µM) and inhibits sarin-induced AChE deactivation (IC50 = 1.21 mM) in hemoglobin-free erythrocyte ghosts. Formulations containing pralidoxime have been used to treat organophosphate poisoning.Formal Name: 2-[(hydroxyimino)methyl]-1-methyl-pyridinium, monochloride. CAS Number: 51-15-0. Synonyms: 2-PAM. Molecular Formula: C7H9N2O . Cl. Formula Weight: 172.6. Purity: >95%. Formulation: (Request formulation change), A crystalline solid. Solubility: DMSO: 1 mg/ml, PBS (pH 7.2): 10 mg/ml. lambdamax: 247, 300 nm. SMILES: C[N+]1=CC=CC=C1/C=N/O.[Cl-]. InChi Code: InChI=1S/C7H8N2O.ClH/c1-9-5-3-2-4-7(9)6-8-10,/h2-6H,1H3,1H. InChi Key: HIGSLXSBYYMVKI-UHFFFAOYSA-N.
| Keywords: | 2-PAM, 2-[(hydroxyimino)methyl]-1-methyl-pyridinium, monochloride |
| Supplier: | Cayman Chemical |
| Supplier-Nr: | 23666 |
Properties
| Application: | Acetylcholinesterase (AChE) reactivator |
| MW: | 172.6 D |
| Formula: | C7H9N2O . Cl |
| Purity: | >95% |
| Format: | Crystalline Solid |
Database Information
| CAS : | 51-15-0| Matching products |
| KEGG ID : | K01049 | Matching products |
Handling & Safety
| Storage: | -20°C |
| Shipping: | +20°C (International: -20°C) |
| Signal Word: | Warning |
| GHS Hazard Pictograms: |
|
| H Phrases: | H302, H312, H315, H319, H332, H335 |
| P Phrases: | P261, P264, P270, P271, P280, P312, P321, P330, P301+P310, P302+P352, P304+P340, P305+P351+P338, P332+P313, P337+P313, P361+P364, P362+P364, P405, P403+P233, P501 |
Caution
Our products are for laboratory research use only: Not for administration to humans!
Our products are for laboratory research use only: Not for administration to humans!
Information about the product reference will follow.
more
You will get a certificate here
Viewed

