If you have any questions, please use our Contact Form.
You can also order by e-mail: info@biomol.com
Larger quantity required? Request bulk
You can also order by e-mail: info@biomol.com
Larger quantity required? Request bulk
Viewed
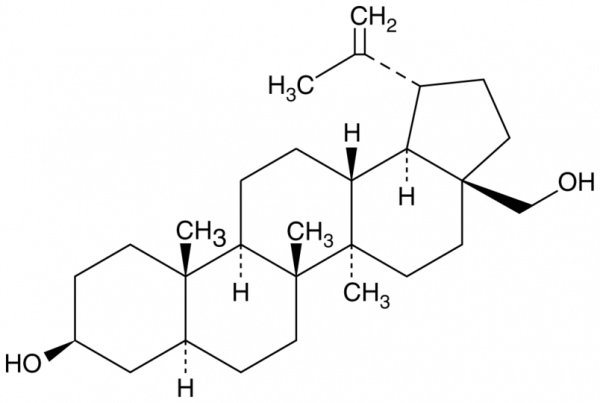
| Keywords: | NSC 4644, Trochol, lup-20(29)-ene-3beta,28-diol |
| Supplier: | Cayman Chemical |
| Supplier-Nr: | 11041 |
Properties
| Application: | Cholesterol control |
| MW: | 442.7 D |
| Formula: | C30H50O2 |
| Purity: | >98% |
| Format: | Crystalline Solid |
Database Information
| CAS : | 473-98-3| Matching products |
| KEGG ID : | K09107 | Matching products |
Handling & Safety
| Storage: | -20°C |
| Shipping: | +20°C (International: -20°C) |
| Signal Word: | Warning |
| GHS Hazard Pictograms: |
|
| H Phrases: | H371 |
| P Phrases: | P260, P264, P270, P273, P308+P311, P405, P501 |
