Cookie preferences
This website uses cookies, which are necessary for the technical operation of the website and are always set. Other cookies, which increase the comfort when using this website, are used for direct advertising or to facilitate interaction with other websites and social networks, are only set with your consent.
Configuration
Technically required
These cookies are necessary for the basic functions of the shop.
"Allow all cookies" cookie
"Decline all cookies" cookie
CSRF token
Cookie preferences
Currency change
Customer-specific caching
FACT-Finder tracking
Individual prices
Selected shop
Session
Comfort functions
These cookies are used to make the shopping experience even more appealing, for example for the recognition of the visitor.
Note
Show the facebook fanpage in the right blod sidebar
Statistics & Tracking
Affiliate program
Conversion and usertracking via Google Tag Manager
Track device being used
| Item number | Size | Datasheet | Manual | SDS | Delivery time | Quantity | Price |
|---|---|---|---|---|---|---|---|
| Cay30586-5 | 5 U | - |
6 - 10 business days* |
720.00€
|
If you have any questions, please use our Contact Form.
You can also order by e-mail: info@biomol.com
Larger quantity required? Request bulk
You can also order by e-mail: info@biomol.com
Larger quantity required? Request bulk
Thioredoxin reductase 1 (TrxR1) is an oxidoreductase encoded by the TXNRD1 gene in humans and a... more
Product information "Thioredoxin Reductase 1 (rat, recombinant, aa 3-499)"
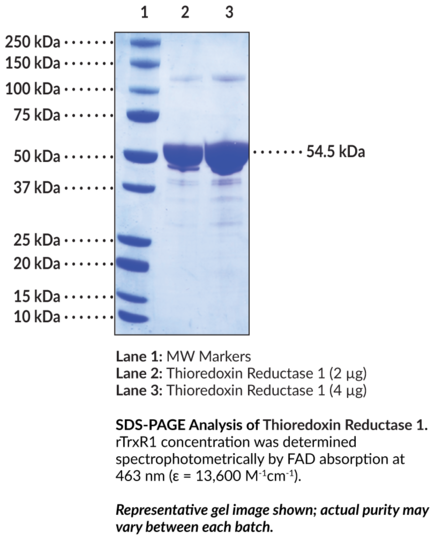
Thioredoxin reductase 1 (TrxR1) is an oxidoreductase encoded by the TXNRD1 gene in humans and a member of the antioxidant thioredoxin system, which is involved in the maintenance of cellular thiol redox homeostasis. It exists as a homodimer and contains a dimer interface domain, FAD and NADPH binding domains, an N-terminal redox catalytic site, and a C-terminal selenocysteine residue, which is essential for the catalytic activity of TrxR1. TrxR1 is ubiquitously expressed, localizes to the cytoplasm, and is regulated by the antioxidant transcription factor Nrf2. TrxR1 catalyzes the NADPH-dependent reduction of oxidized thioredoxin (Trx), restoring the disulfide reductase function of Trx, which regulates redox-sensitive transcription factors, such as NF-kappaB and p53, and has roles in apoptosis and cell signaling. Genome-wide deletion of Txnrd1 is embryonic lethal in mice. Increased serum TrxR1 activity is associated with reduced progression-free survival in patients with non-small cell lung cancer (NSCLC). Cayman's Thioredoxin Reductase 1 (rat, recombinant, aa 3-499) protein can be used for enzyme activity assay and Western blot (WB) applications.Synonyms: NADPH-dependent Thioredoxin Reductase, TrxR1, Txnrd1. Purity: >45% estimated by SDS-PAGE. Source: Active recombinant rat TrxR1 expressed in E. coli. Amino Acids: 3-499. MW: 54.5 kDa. Formulation: (Request formulation change), 50 mM Tris, pH 7.4, with 800 mM sodium chloride and 10% glycerol. Applications: Enzyme activity assay and Western blot (WB). Specific Activity: batch specific. UniProt Accession #: O89049.
| Keywords: | TR, TR, Trxr1, Txnrd1, Thioredoxin reductase TR1, Thioredoxin reductase TR1, Thioredoxin reductase 1, cytoplasmic, Thioredoxin reductase 1, cytoplasmic, NADPH-dependent thioredoxin reductase, NADPH-dependent thioredoxin reductase |
| Supplier: | Cayman Chemical |
| Supplier-Nr: | 30586 |
Properties
| Application: | Enzyme activity assay, WB |
| Conjugate: | No |
| Purity: | >50% estimated by SDS-PAGE |
| Format: | Purified |
Database Information
| UniProt ID : | O89049 | Matching products |
Handling & Safety
| Storage: | -80°C |
| Shipping: | -80°C (International: -80°C) |
| Signal Word: | Warning |
| GHS Hazard Pictograms: |
|
| H Phrases: | H373, H316 |
| P Phrases: | P260, P314, P332+313, P501 |
Caution
Our products are for laboratory research use only: Not for administration to humans!
Our products are for laboratory research use only: Not for administration to humans!
Information about the product reference will follow.
more
You will get a certificate here
Viewed

