Cookie preferences
This website uses cookies, which are necessary for the technical operation of the website and are always set. Other cookies, which increase the comfort when using this website, are used for direct advertising or to facilitate interaction with other websites and social networks, are only set with your consent.
Configuration
Technically required
These cookies are necessary for the basic functions of the shop.
"Allow all cookies" cookie
"Decline all cookies" cookie
CSRF token
Cookie preferences
Currency change
Customer-specific caching
FACT-Finder tracking
Individual prices
Selected shop
Session
Comfort functions
These cookies are used to make the shopping experience even more appealing, for example for the recognition of the visitor.
Note
Show the facebook fanpage in the right blod sidebar
Statistics & Tracking
Affiliate program
Conversion and usertracking via Google Tag Manager
Track device being used
| Item number | Size | Datasheet | Manual | SDS | Delivery time | Quantity | Price |
|---|---|---|---|---|---|---|---|
| Cay43211-100 | 100 µg | - |
6 - 10 business days* |
516.00€
|
If you have any questions, please use our Contact Form.
You can also order by e-mail: info@biomol.com
Larger quantity required? Request bulk
You can also order by e-mail: info@biomol.com
Larger quantity required? Request bulk
Peroxiredoxin-6 (Prx6) is a 1-Cys-containing Prx that is involved in the maintenance of cellular... more
Product information "Peroxiredoxin-6 (human, recombinant)"
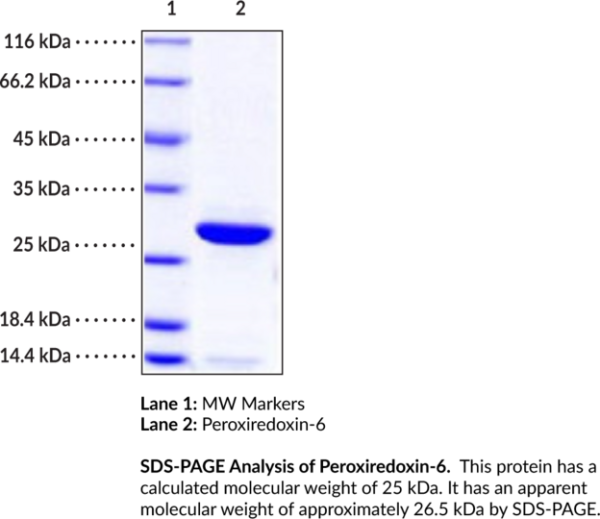
Peroxiredoxin-6 (Prx6) is a 1-Cys-containing Prx that is involved in the maintenance of cellular thiol redox homeostasis and phospholipid metabolism. It exists as a homodimer and is composed of a thioredoxin fold-containing catalytic domain, which contains a peroxidatic cysteine (Cp), and a C-terminal arm. Unlike 2-Cys-containing Prxs, Prx6 does not contain a resolving cysteine (Cr) and uses glutathione (GSH) instead. Prx6 is ubiquitously expressed and localizes to the cytosol, nucleus, mitochondria, and plasma membrane. It has three enzymatic activities: glutathione peroxidase (GPX), acidic calcium-independent phospholipase A2 (aiPLA2), and lysophosphatidylcholine acyltransferase (LPCAT). The LPCAT activity, which selectively uses palmitoyl-CoA as a substrate, is coupled to the aiPLA2 activity and is selective for lysophosphatidylcholine over lysophosphatidylethanolamine, lysophosphatidylglycerol, lysophosphatidylinositol, and lysophosphatidylserine. These activities, coupled with the GPX activity, provide Prx6 the ability to repair oxidized cell membranes. Prx6 is also involved in the activation of NADPH oxidase 2 (NOX2). Tumor levels of Prx6 are increased in patients with non-small cell lung cancer (NSCLC) and serum levels of Prx6 are decreased in patients with asthma. Cayman's Peroxiredoxin-6 (human, recombinant) protein consists of 235 amino acids and has a calculated molecular weight of approximately 25 kDa. By SDS-PAGE, under reducing conditions, the apparent molecular mass of the protein is 26. kDa.. Protein function: Thiol-specific peroxidase that catalyzes the reduction of hydrogen peroxide and organic hydroperoxides to water and alcohols, respectively (PubMed:9497358, PubMed:10893423). Can reduce H(2)O(2) and short chain organic, fatty acid, and phospholipid hydroperoxides (PubMed:10893423). Also has phospholipase activity, can therefore either reduce the oxidized sn-2 fatty acyl group of phospholipids (peroxidase activity) or hydrolyze the sn-2 ester bond of phospholipids (phospholipase activity) (PubMed:10893423, PubMed:26830860). These activities are dependent on binding to phospholipids at acidic pH and to oxidized phospholipds at cytosolic pH (PubMed:10893423). Plays a role in cell protection against oxidative stress by detoxifying peroxides and in phospholipid homeostasis (PubMed:10893423). Exhibits acyl-CoA-dependent lysophospholipid acyltransferase which mediates the conversion of lysophosphatidylcholine (1-acyl-sn-glycero-3- phosphocholine or LPC) into phosphatidylcholine (1,2-diacyl-sn-glycero- 3-phosphocholine or PC) (PubMed:26830860). Shows a clear preference for LPC as the lysophospholipid and for palmitoyl CoA as the fatty acyl substrate (PubMed:26830860). [The UniProt Consortium]
| Keywords: | AOP2, PRDX6, NSGPx, aiPLA2, LPCAT-5, 1-Cys PRX, 24 kDa protein, Peroxiredoxin-6, 1-Cys peroxiredoxin, Liver 2D page spot 40, Antioxidant protein 2, LPC acyltransferase 5, Lyso-PC acyltransferase 5, Red blood cells page spot 12 |
| Supplier: | Cayman Chemical |
| Supplier-Nr: | 43211 |
Properties
| Conjugate: | No |
| Host: | E.coli |
| Purity: | >95% (SDS-PAGE) |
| Format: | Lyophilized |
Database Information
| KEGG ID : | K11188 | Matching products |
| UniProt ID : | P30041 | Matching products |
| Gene ID : | GeneID 9588 | Matching products |
Handling & Safety
| Storage: | -80°C |
| Shipping: | -80°C (International: -80°C) |
Caution
Our products are for laboratory research use only: Not for administration to humans!
Our products are for laboratory research use only: Not for administration to humans!
Information about the product reference will follow.
more
You will get a certificate here
Viewed

