Cookie preferences
This website uses cookies, which are necessary for the technical operation of the website and are always set. Other cookies, which increase the comfort when using this website, are used for direct advertising or to facilitate interaction with other websites and social networks, are only set with your consent.
Configuration
Technically required
These cookies are necessary for the basic functions of the shop.
"Allow all cookies" cookie
"Decline all cookies" cookie
CSRF token
Cookie preferences
Currency change
Customer-specific caching
FACT-Finder tracking
Individual prices
Selected shop
Session
Comfort functions
These cookies are used to make the shopping experience even more appealing, for example for the recognition of the visitor.
Note
Show the facebook fanpage in the right blod sidebar
Statistics & Tracking
Affiliate program
Conversion and usertracking via Google Tag Manager
Track device being used
| Item number | Size | Datasheet | Manual | SDS | Delivery time | Quantity | Price |
|---|---|---|---|---|---|---|---|
| 002-0102-0002 | 2 mg | - | - |
6 - 10 business days* |
204.00€
|
If you have any questions, please use our Contact Form.
You can also order by e-mail: info@biomol.com
Larger quantity required? Request bulk
You can also order by e-mail: info@biomol.com
Larger quantity required? Request bulk
Cat IgG whole molecule was prepared from normal serum by a multi-step process which includes... more
Product information "CAT IgG whole molecule"
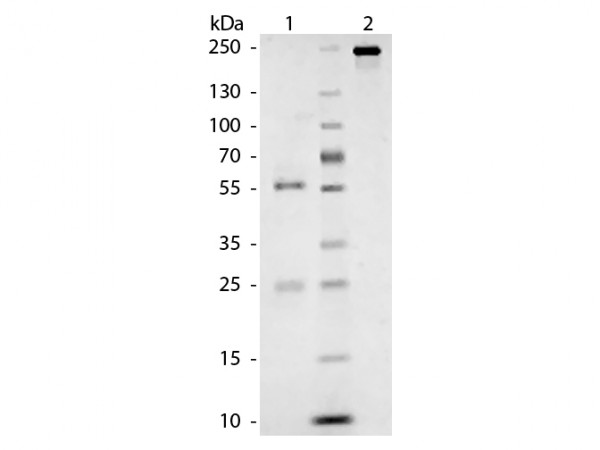
Cat IgG whole molecule was prepared from normal serum by a multi-step process which includes delipidation, salt fractionation and ion exchange chromatography followed by extensive dialysis against the buffer stated above. Cat IgG whole molecule assayed by immunoelectrophoresis resulted in a single precipitin arc against anti-Cat IgG and anti-Cat Serum. Secreted as part of the adaptive immune response by plasma B cells, immunoglobulin G constitutes 75% of serum immunoglobulins. Immunoglobulin G binds to viruses, bacteria, as well as fungi and facilitates their destruction or neutralization via agglutination (and thereby immobilizing them), activation of the compliment cascade, and opsinization for phagocytosis. The whole IgG molecule possesses both the F(c) region, recognized by high-afinity Fc receptor proteins, as well as the F(ab) region possessing the epitope-recognition site. Both heavy and light chains of the antibody molecule are present. Cat IgG whole molecule is ideal for researchers in Cancer, Immunology, and Cell Biology.
| Supplier: | Rockland Immunochemicals |
| Supplier-Nr: | 002-0102-0002 |
Properties
| Application: | ELISA, IHC, WB |
| Conjugate: | No |
| Host: | Cat |
| Format: | Purified |
Database Information
Handling & Safety
| Storage: | +4°C |
| Shipping: | +20°C (International: +20°C) |
Caution
Our products are for laboratory research use only: Not for administration to humans!
Our products are for laboratory research use only: Not for administration to humans!
Information about the product reference will follow.
more
You will get a certificate here
Viewed