Cookie-Einstellungen
Diese Website benutzt Cookies, die für den technischen Betrieb der Website erforderlich sind und stets gesetzt werden. Andere Cookies, die den Komfort bei Benutzung dieser Website erhöhen, der Direktwerbung dienen oder die Interaktion mit anderen Websites und sozialen Netzwerken vereinfachen sollen, werden nur mit Ihrer Zustimmung gesetzt.
Konfiguration
Technisch erforderlich
Diese Cookies sind für die Grundfunktionen des Shops notwendig.
"Alle Cookies ablehnen" Cookie
"Alle Cookies annehmen" Cookie
Ausgewählter Shop
CSRF-Token
Cookie-Einstellungen
FACT-Finder Tracking
Individuelle Preise
Kundenspezifisches Caching
Session
Währungswechsel
Komfortfunktionen
Diese Cookies werden genutzt um das Einkaufserlebnis noch ansprechender zu gestalten, beispielsweise für die Wiedererkennung des Besuchers.
Facebook-Seite in der rechten Blog - Sidebar anzeigen
Merkzettel
Statistik & Tracking
Endgeräteerkennung
Kauf- und Surfverhalten mit Google Tag Manager
Partnerprogramm
| Artikelnummer | Größe | Datenblatt | Manual | SDB | Lieferzeit | Menge | Preis |
|---|---|---|---|---|---|---|---|
| 012-0107 | 1 mg | - | - |
6 - 10 Werktage* |
344,00 €
|
Bei Fragen nutzen Sie gerne unser Kontaktformular.
Bestellen Sie auch per E-Mail: info@biomol.com
Größere Menge gewünscht? Bulk-Anfrage
Bestellen Sie auch per E-Mail: info@biomol.com
Größere Menge gewünscht? Bulk-Anfrage
Rat IgM whole molecule was prepared from normal serum by a multi-step process which includes... mehr
Produktinformationen "Rat IgM, whole molecule"
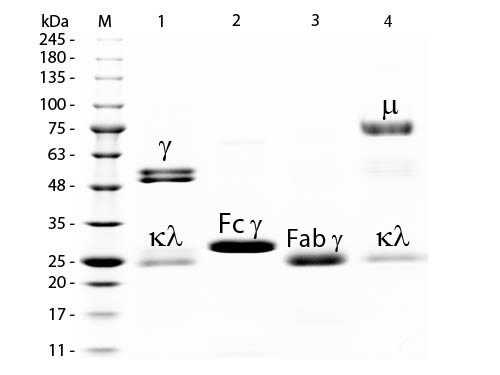
Rat IgM whole molecule was prepared from normal serum by a multi-step process which includes delipidation, selective precipitation and tandem molecular sieve chromatography followed by extensive dialysis against the buffer stated above. Rat IgM whole molecule assayed by immunoelectrophoresis resulted in a single precipitin arc against anti-Rat Serum and anti-Rat IgM (µ chain specific). No reaction was observed against anti-Rat IgG F(c). Some light chain cross-reactivity will occur with anti-Rat IgG. Immunoglobulin M is the largest antibody isotype and the first to be secrected against an initial exposure to antigen. IgM is predominantly produced in the spleen. Formed from covalently linking 5 immunoglobulins together, the approixmate molecular weight of IgM is 900kDa and possesses 10 binding sites (though due to the size of most antigens, not all sites are capable of binding at once). Due to this large size, IgM is typically isolated to the serum.
| Hersteller: | Rockland Immunochemicals |
| Hersteller-Nr: | 012-0107 |
Eigenschaften
| Anwendung: | ELISA, IHC, WB |
| Konjugat: | No |
| Wirt: | Rat |
| Format: | Purified |
Datenbank Information
Handhabung & Sicherheit
| Lagerung: | -20°C |
| Versand: | +4°C (International: +4°C) |
Achtung
Nur für Forschungszwecke und Laboruntersuchungen: Nicht für die Anwendung im oder am Menschen!
Nur für Forschungszwecke und Laboruntersuchungen: Nicht für die Anwendung im oder am Menschen!
Hier folgen Informationen zur Produktreferenz.
mehr
Hier kriegen Sie ein Zertifikat
Loggen Sie sich ein oder registrieren Sie sich, um Analysenzertifikate anzufordern.
Bewertungen lesen, schreiben und diskutieren... mehr
Kundenbewertungen für "Rat IgM, whole molecule"
Bewertung schreiben
Loggen Sie sich ein oder registrieren Sie sich, um eine Produktbewertung abzugeben.
Zuletzt angesehen